Rejuvapen NXT®
Description
The NXT Evolution
Microneedling System
FDA 510k Cleared
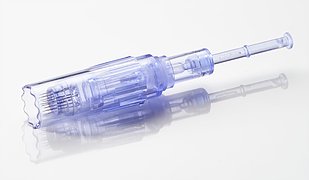
The Rejuvapen System utilizes an array of stainless steel needle cartridges. This innovative new technology can help improve your appearance with little recovery time. As a result, the Rejuvapen is a perfect noninvasive lunchtime procedure that delivers exceptional results.
- 12 stainless steel tips
- Market-leading torque
- Designed and manufactured in the USA
- Durable medical-grade aluminum design
- Scalloped edge tip eliminates suction and clogging
- Plug-in system for full power treatments and consistent output
- Ergonomic and lightweight design for enhanced comfort
ISO Tips™
Patent Pending seal creates a barrier between tip and device.
Smart Touch
Digital fingertip control of on/off and 9 separate speeds.
Infinite Dial
For precise and customizable treatments, 0.02-2.5mm.
Dedicated to Safety
The Rejuvapen NXT continues to demonstrate its commitment to safety, quality, and excellence through safety testing. As a market leader, the Rejuvapen has performed over 500,000 safe and effective treatments.
- Life Cycle Testing (Cleaning & Disinfection)
- ISO Seal Integrity Testing and Process Validation
- Biocompatibility
- CE Mark
AAMI / IEC 60601-1-2: 2004 Electromagnetic Compatibility, Immunity, Emissions, and Essential Performance Testing
EN 60601-1: 2006 + A1: 2013 Electrical Safety Testing
ES 60601-1: 2005 + C1 (2009) + A1 (2010) + A2 (2010) Electrical Safety Testing
FDA 510k Cleared: K192138
Tested for Quality and Safety
FDA 510k Cleared and Trusted by thousands of Aesthetic Practitioners
Quick Processing and Ship Times
Schedule a Demo Today!
Contact Us
We’re ready to lead you into the future of Aesthetics.




Reviews
There are no reviews yet.